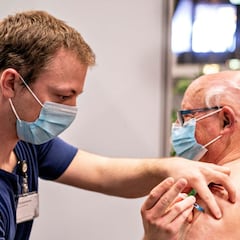
What is a cerebral venous sinus thrombosis? Blood clot symptons and causes
With the covid-19 vaccine rollout picking up steam in the US and around the world, some cocerns have been raised over thrombotic complications.
As vaccine rollout continues in the United States and around the world, concerns over possible side effects have some concerned.
Cerebral Venous Sinus Thrombosis and covid-19 vaccination
While the fatal side effects of vaccines have been widely covered in the media, it is important to note that deadly outcomes are extremely rare and as yet it is not understood if there is indeed an actual link between the vaccine and a subsequent incident, nor the mechanism by which this could occur.
- Covid-19 and US vaccine rollout: live updates
- How many J&J vaccines have been administered in the US?
- Differences between the Pfizer, Moderna and J&J/Janssen covid-19 vaccines
- US stimulus checks: latest news
- What to do if you get covid-19 between vaccine doses?
Individuals who forego vaccinations are much more likely to risk death by covid-19 than by getting vaccinated.
What are some of the side effects, and how common are they?
One of the emerging side effects that has been linked to the Johnson and Johnson (J&J) vaccine is Cerebral Venous Sinus Thrombosis or CVST. According to Johns Hopkins Medicine, CVST is a condition wherein a blood clot that forms in the brain that prevents blood from draining and can cause headaches, blurred vision, fainting or loss of consciousness, loss of control over movement, seizures and, coma. CVST can be treated and although it can be deadly, the condition only affects about 5 in 1,000,000 people each year.
CVST has received more attention since the large-scale rollout of the J&J vaccine but of the more than 7 million people inoculated, six people have developed the condition. Of the six patients, one case has been fatal, and one continues to be hospitalized in critical condition.
Today, the FDA/CDC halted the administration of the J&J vaccine due to concern for cerebral venous sinus thrombosis. I looked into risk in general - and here’s a simple graphic. pic.twitter.com/SHSU1IHeeV
— Jesse O’Shea MD, MSc (@JesseOSheaMD) April 13, 2021
It is not to say that fears about CVST are not legitimate, but this is a fatality rate far less than one percent. Meanwhile, on April 12th, nearly 1,000 people in the United States died due to Covid-19.
What steps are being taken to protect the public?
A joint press call held on Tuesday between the Principal Deputy Director at the Center for Disease Control and Prevention (CDC), Dr. Anne Schuchat, and Director for Biologics Evaluation and Research at the Food and Drug Administration (FDA) Dr. Mark Peters. On the call, the two experts provided updates on the patient profiles of those impacted, responded to questions from the media, and outlined what steps were being taken by both organizations to protect the public.
Recently, you might have heard the term CVST in connection with coverage around the Covid-19 vaccine. #CVST occurs when a blood clot forms in the brain's venous sinuses. This prevents blood from draining out of the brain. This is an extremely rare condition. #WTDay21 #bloodclot pic.twitter.com/TFowO5pjue
— World Thrombosis Day (@thrombosisday) April 14, 2021
Related stories
Dr. Peters stated that the six patients who developed CVST were women between the ages of 18 and 48 and that “out of an abundance of caution,” both organizations are recommending that the J&J vaccine rollout be “paused” until more information is gathered. Reiterating comments made by Dr. Schuchat, Dr. Peters reminded those on the call that “Right now these events seem to be extremely rare” and that the federal government takes all reports of “adverse events after vaccinations very seriously”. In the coming days, both organizations will update the information available to health care providers on the J&J vaccines to help them make more informed decisions on which vaccinations to provide their patients.
Moving forward with vaccinations in the United States
While these side effects may create skepticism among the population, it is critical to look at the big picture. With nearly 122,000,000 million people in the United States being partially or fully vaccinated, these adverse cases make up a tiny percentage of total outcomes. The risk of dying or developing life-long complications from covid-19 poses a much greater threat to nearly three-quarters of the US population who have yet to receive a vaccine.